Environment & Energy
Related: About this forumThe Biogeochemical Fate of the Long Lived Radioactive Fission Product Selenium-79.
The paper to which I'll refer in this post is this one: The Biogeochemical Fate of Se(VI) in Bentonite Systems Relevant to the Disposal of High-Level Radioactive Waste Max Rose, Samuel Shaw, Katherine Morris, Christopher Boothman, Matthew Bailey, Dirk Engelberg, and Jonathan R. Lloyd Environmental Science & Technology 2026 60 (5), 4124-4133.
I have some disagreements with the contents of the paper, beginning with the claim that we should dispose of so called "nuclear waste at all. I don't actually consider any component of used nuclear fuel - and I'm fairly familiar with all of them - to be "waste." I regard the word "product" in the term "fission products" to be a pun. To my mind fission products are valuable, but perhaps one of them, that discussed in this article 79Se could be problematic, in a way. Let's look at it a little deeper.
Let me quote from the paper's introduction to bring forth another bone of contention:
The italicized and bolded term "high yield" is wrong. The yield of 79Se is low compared to other fission products, almost to the point of triviality, which, as I will argue below, is somewhat unfortunate. The stable isotope associated with mass number 79 is 79Br, one of the two stable isotopes of bromine. Below are graphics, from the Brookhaven National Lab (BNL) Nuclear Data pages, showing the distribution of fission products for the two major fission sources found in a commercial light water or heavy water reactor with a mass number abscissa, 235U and 239Pu, the latter generated in situ in operations from neutron capture in the fertile but not critically fissionable isotope 238U.
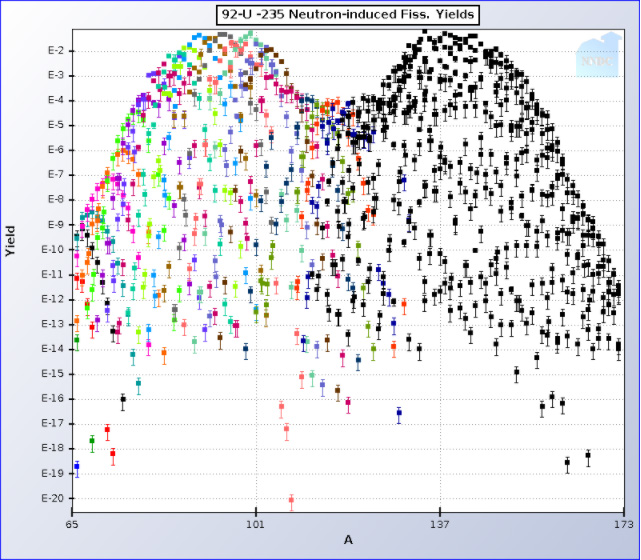
235U:
239Pu:
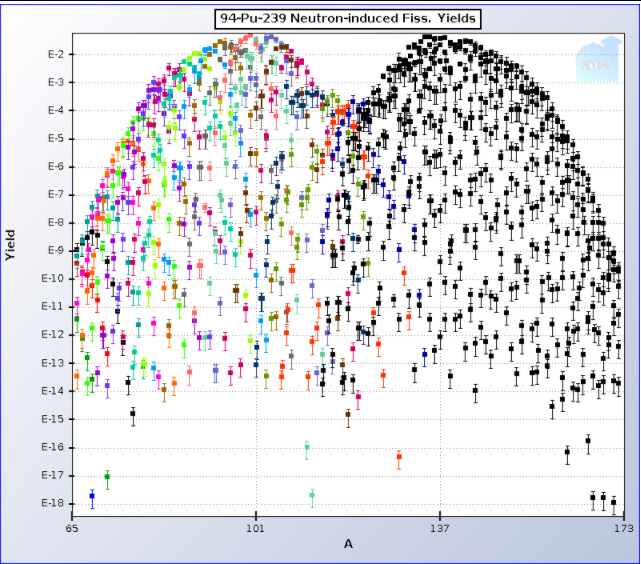
It is clear by using the logarithmic ordinate that the mass number in both cases at 79 amu, is at best a minor constituent of used nuclear fuel. In the case of uranium fission the initial elements initially formed at mass number 79 are 79Zn, 79Ga 79Ge, 79As, and finally 79Se, respectively highly radioactive and thus short lived isotopes, of zinc, gallium, germanium, and arsenic are elements in the decay chain leading up to 79Se which has a half-life of 3.27 X 105 years. Only only tiny traces of the stable non-radioactive naturally occurring isotope of bromine 79Br will have time to form. (The result of the long half life of 79Se is that bromine found in used nuclear fuel is essentially monoisotopic 81Br, which makes it unique for isotopic labeling experiments for example in studying degradation mechanisms of toxic and persistent brominated flame retardants.) The Brookhaven data pages give the graphic data shown above numerically. The sum of the fractional yield of all fission products of mass number 79 can be shown, using this data for 235U to be 4.47 X 10-4 (0.047%) and for 239Pu, 2.29 X 10-4 (In asymmetric fission the left “hump” moves to the right as the atomic weight of fissioning nuclei increases, so that by the time isotopes of fermium, element 100, are encountered, the distribution is approximately a single “hump,” the fission product distribution is essentially symmetric.)
One can estimate from these figures how much 79Se is available for recovery from used nuclear fuel in the United States, variously reported as somewhere, after approximately 70 years of commercial nuclear power in the United States, at approaching around 90,000 metric tons. At modern burn ups about 95% of used nuclear fuel remains as unchanged uranium, 1% is plutonium with small amounts of neptunium and the higher actinides such as americium (depending on cooling time) and traces of curium, berkelium and californium. About 4% is fission products, suggesting about 3,600 tons. If we assume, ignoring some variances, that about half of the energy produced by nuclear fuel comes from 235U and half comes from 239Pu formed in situ – again a rough approximation, the yield of 79Se should be somewhere around 3.4 X 10-4, the US supply, ignoring decay because of the long half life, should be around 800 kg, collected over approximately 70 years. Fission will result in the formation of several of the stable and metastable natural isotopes of selenium, for example 76Se. 77Se, 78Se and 80Se as well as 82Se. The naturally occurring latter isotope has been discovered to be radioactive, but very weakly so, as its half-life exceeds the age of the universe. The total fractional mixture of isotopes of Selenium for uranium (235U) thermal spectrum fission, using fission yield data available at BNL, amounts to 5.27 X 10-3. For plutonium, 239Pu , the quantity is less, 1.03 X 10-3. This suggests that the roughly 800 kg 79Se will be suspended in roughly 5600 kg (5.6 tons) of nonradioactive selenium; about 14% will be the long lived radioactive isotope of interest. Elemental selenium has three allotropes having different densities, and the selenium coming out of used nuclear fuel will differ in its atomic weight from natural selenium because of its non-stellar fission source. However, as this is a “back of the envelope” calculation we can estimate the volume of isolated selenium by taking an “average” density of 4.4 g/cm3, close to the density of the alpha allotrope. This suggests that the US supply of fissiogenic selenium could fit in a little over 1.2 cubic meters, a box 106 centimeters on a side, which could easily fit into the bed of a pickup truck..
It is rather difficult to imagine that it would be harder to contain 1.2 cubic meters of anything indefinitely than it would be to contain the 37 billion tons of gaseous carbon dioxide, the dangerous fossil fuel waste, being released, at the current rate, each year by the use of dangerous fossil fuels.
There are seven fission products commonly found in used nuclear fuel that have particularly long half-lives, over 100,000 years. They are the aforementioned 79Se, (t1/2 = 327,000 years,) 93Zr (zirconium) (t1/2 = 1,600,000 years), 99Tc (technetium) (t1/2 = 211,100 years), 107Pd (palladium) (t1/2 = 6,100,000 years), 126Sn, (tin) (t1/2 = 230,000 years), 129I, (iodine) (t1/2 = 15,900,000 years) and 135Cs (cesium) (t1/2 = 1,300,000 years). To my mind, all of these isotopes have potentially useful applications, in several cases in nuclear applications. Technetium can replace its extremely rare but extremely valuable naturally occurring congener, rhenium in almost every application, for example, for the production of machinable tungsten alloys. (The world supply of technetium can easily be made to exceed that of rhenium.)
Of these isotopes, only one, the isotope of tin, 126Sn, is a strong emitter of highly penetrating gamma rays. Of course, if one thinks of this properly, one can recognize that gamma radiation is a particularly wonderful thing, since it can do things few other things can do, for instance cleave the strong carbon fluorine bond featured in atmospheric HFCs, and for that matter, water polluted with the otherwise intractable perfluoro organic substances, (PFOS) which are now generating enormous concern in the environmental community.
For this reason, the gamma emissions of 126Sn, along with the presence of selenium’s isotopes in used nuclear fuel including the long lived 79Se, which always comes out of used nuclear fuel with stable isotopes (as does tin), intrigue me. The reason can be found in this paper: (Insert RSC paper)
The main impediment to utilizing these isotopes rather than attempting to dump them somewhere, although the risk of nuclear dumps is vanishingly small, is economic, specifically that there isn’t very much of these materials available. Thus the scale at which they might operate is limited. The environmental superiority of nuclear energy to all other forms of primary energy is closely tied to the fact that the yield of these isotopes is very small. This said, an argument can, and should, be made.
It is important to note that the longer the half-life of a nucleon is, the less radioactive it is. Uranium’s main isotope, 238U, has a half-life that is roughly about the same as what is generally taken to be the lifetime of the Earth, about 4.5 billion years. Anyone who has a granite kitchen countertop is likely to have uranium in it, and yet people are not killed, nor their health impaired, by radiation from their countertop, nor by swimming in the ocean, which contains over 4 billion tons of uranium.
Because of historical practices in nuclear fuel reprocessing, as well as nuclear weapons testing most people on Earth have small amounts of highly mobile129I (iodine) in their thyroids, but thyroid cancer is rare compared to other cancers, notably that caused by dangerous fossil fuel waste (aka “air pollution” ), lung cancer. Rates of thyroid cancer in men does not appear in the top ten, in women, it is the eighth most common cancer, and in any case, thyroid cancer is rarely fatal, with cure rates exceeding 99%.
Antinukes and “I’m not an antinuke” antinukes around here like to carry on all the time about Fukushima, where the death toll from radiation was miniscule, if not zero, whereas the death toll from the Sendai earthquake natural disaster that destroyed the reactors along with a coastal city, was roughly 20,000, suggesting that coastal cities are more dangerous than breached melted nuclear reactors. For a period of time, in Japan, people shut well functioning nuclear reactors, thus killing people with fossil fuel waste, but there was no movement to ban coastal cities.
This is a point I make repeatedly in this space and elsewhere, living in coastal city is more dangerous than living next to a nuclear plant with melting fuel and a breached containment building.
Go figure.
Anyway. Let us turn to one of the 7 “long lived” fission products, the gamma emitting isotope of tin, 126Sn. Tin it turns out, forms a binary compound with selenium, SnSe, which is completely insoluble in water. It has an interesting property described in the following paper:
Zhao, Li-Dong and Chang, Cheng and Tan, Gangjian and Kanatzidis, Mercouri G. SnSe: a remarkable new thermoelectric material Energy Environ. Sci., 2016,9, 3044-3060.
A thermoelectric device is a device that converts heat directly into electricity with no moving parts. Devices of this type are not new; the Voyager spacecraft (among many other space devices) which was launched nearly half a century ago are still sending signals to Earth having left the solar system. The above paper reports on the properties of pure tin selenide and was published in 2016, about a decade ago. Further refinements of this system are the subject of one of my posts in 2022:
High ZT thermoelectric material by doping SnTe with copper and selenium.
From that text:
The thermal efficiency of an RTG is given by a formula utilizing a dimensionless parameter called "ZT" (it is not a product of two variables) according to the following equation:
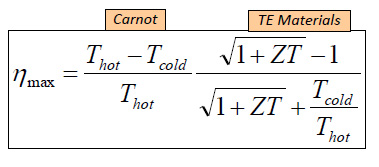
ZT is determined by the following formula:
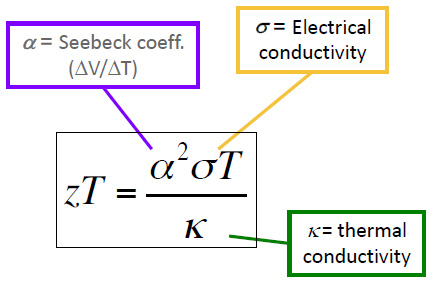
where S, σ, T, and κ are the Seebeck coefficient, (a property of the material) electrical conductivity, absolute temperature, and thermal conductivity, respectively. Note that the value of ZT requires that two variables which normally trend together, electrical conductivity and thermal conductivity - metals usually possess high values of both - must trend in opposite directions to maximize ZT.
The "State of the Art" RTG utilized on Curiosity is the MMRTG (Multimission RTG).
The value of the MMRTG is shown in the following graphic from a 2019 slide show given by Dr. Giacomo Cerretti, a post doc at JPL. The following is also from his slide show, showing where the MMRTG lies in terms of ZT and efficiency.
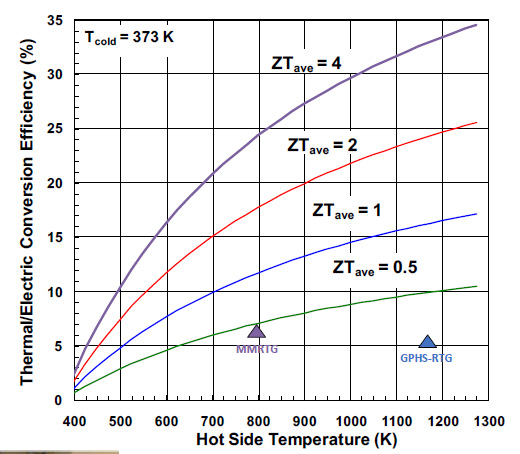
The MMRTG's thermoelectric material is lead telluride.
Right now, most of the used nuclear fuel in the United States is sitting in casks on the sites where it was generated. If one looks around the internet, one can find pictures of nuclear advocates hugging these casks to show that they are not particularly dangerous.
I find this containment in casks to be unfortunate; I would rather that the materials within them should be utilized including use to irradiate the air, since radiation has the effect of destroying certain classes of greenhouse gases other than carbon dioxide, notably residual CFC's that have survived since the signing of the Montreal Protocol as well as that present from cheating, the replacement HFCs which have very high warming potentials compared to carbon dioxide, as well as the ubitiquous contaminant that of growing concern, trifluoroacetic acid, the ultimate degradant of PFAS, as well as other volatile PFAS themselves.
What better way could there be to build casks designed to let air flow over a radioactive thermoelectric device containing both radioactive tin and radioactive selenium?
Recently I have been putting together some notes for my son on the idea of using transplutonium actinides, in particular americium, as nuclear fuels. Such use would be expected to produce curium isotopes, in particular 242Cm and 244Cm that demonstrate higher thermal output than traditional 238Pu plutonium isotope. (The former curium isotope decays to 238Pu.) I might add that there are many other fission products that might generate heat over long periods in thermoelectric devices. With these as a heat source, and the SnSe (doped or otherwise) material generating γ rays, these could provide continuous power at nuclear plants to provide uninterruptable power. I note that had the famous reactors at Fukushima's cooling pumps been powered with thermoelectric backup rather than diesel generators, they would have functioned quite nicely even inundated with seawater. As used nuclear fuel is already safely contained on site, surely thermoelectric devices could be similarly shielded in what would be essentially casks with free air flow.
Again, the limitation is that we don't really have enough radioactive tin and radioactive selenium to make this a broadly available technology, but in a better world, one where we were providing hundreds of Exajoules of nuclear energy as opposed to the 31 Exajoules now being produced, it could become a large scale undertaking, one that would have the effect of cleaning the air while operating.
Burying radioselenium, radiotin, or any component of used nuclear fuel strikes me as a dumb idea, if one has a shred of imagination. If one lacks an imagination of course, one can simply chant nonsense about "dangerous nuclear waste" even though - and I've tested this here many times with "honest to God" antinukes and the less than honest "I'm not an antinuke" antinukes - that there is no evidence, none, that the 70 year history of storing commercial used nuclear fuels has not killed anything like the number of people who will die in the next two hours from dangerous fossil fuel waste, aka air pollution, about 1600 people.
Hence, I think, with due respect to authors cited at the outset, that we really shouldn't give a rat's ass about the biogeochemical fate of fissiogenic selenium.
Nuclear energy is not only the safest form of industrial scale primary energy; it is also the cleanest form of industrial scale energy.
I trust you'll have a pleasant weekend.